|
The end point of a potassium dichromate(VI) titration isn't as easy to see as the end point of a potassium manganate(VII) one. 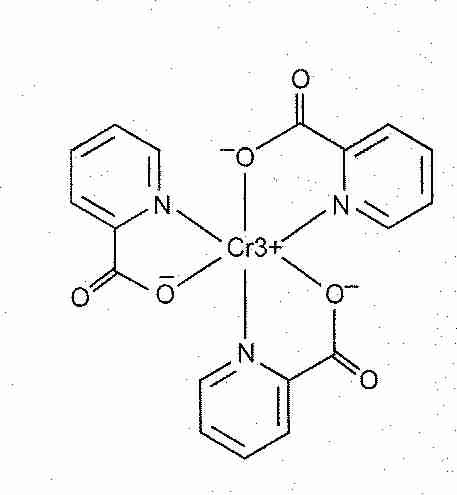
However, the color is made difficult by the strong green also present. This gives a violet-blue color in the presence of excess potassium dichromate(VI) solution. 
There are several such indicators - such as diphenylamine sulfonate. These change color in the presence of an oxidising agent. What is the chemical form for chromium (III) acetate Chemistry Ionic Bonds Writing Ionic Formulas. With potassium dichromate(VI) solution you have to use a separate indicator, known as a redox indicator. C6H9CrO6 Do you mean the chemical formula C6H9CrO6. Unfortunately potassium dichromate(VI) solution turns green as you run it into the reaction, and there is no way you could possibly detect the color change when you have one drop of excess orange solution in a strongly colored green solution. As soon as you add as much as one drop too much, the solution becomes pink - and you know you have reached the end point. As you run the potassium manganate(VII) solution into the reaction, the solution becomes colorless. Chromium (III) Carbonate: Cr 2 (CO 3) 3: Chromium (III) Sulfite: Cr 2 (SO 3) 3: Chromium (III) Sulfate: Cr 2 (SO 4) 3: Chromium (III) Thiosulfate: Cr 2 (S 2 O 3) 3: Chromium (III) Peroxide: Cr 2 (O 2) 3: Chromium (III) Chromate: Cr 2 (CrO 4) 3: Chromium (III) Dichromate: Cr 2 (Cr 2 O 7) 3: Chromium (III) Hydrogen Phosphate: Cr 2 (HPO 4) 3. Potassium manganate(VII) titrations are self-indicating.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
